So you think you know all about Botox®?
So you think you know all about Botox®?
Love it or hate it, the world’s most popular anti-ageing injection treatment certainly has achieved a high level of awareness over the last few years. Speculation as to which celebrities use it (and which don’t) is rife in the media, yet botox is not just for the elite – it is popular among men and women from all walks of society, who choose it for the efficacy with which it can smooth out lines and wrinkles.
Botox is the trade name for botulinum toxin, which is a purified protein derived from the bacterium Clostridium Botulinum.
It has been around for longer than you might think – the bacterium itself was identified in 1895 over 100 years ago. Yet scientists are still discovering more about the powerful toxin which is used to treat lines and wrinkles as well as a host of other medical purposes. Researchers are still learning more about its potential uses in medicine.
Here’s a brief rundown of the history of botox as we know it today:
History of Botox®
| 1895 | The bacterium Bacillus botulinum (later renamed Clostridum botulinum) was identified by Prof. Emile Pierre van Ermengem, of Ellezelles, Belgium. |
| 1920s | Botulinum Toxin Type A was isolated in purified form as a stable acid precipitate at the University of California, San Francisco. |
| 1946 | Researchers succeeded in purifying Botulinum Toxin Type A in crystalline form, providing scientists with the raw material necessary to study the molecule in greater detail. |
| 1950s | Scientists discover that botulinum toxin, injected into a hyperactive muscle, blocks the release of acetylcholine from motor nerve endings, thus inducing a temporary reduction in the targeted muscle’s activity. |
| 1960s/ 1970s | Studies explore botulinum toxin as a treatment for muscle disorders; research into using botox for strabismus (crossed eyes) as an alternative to surgery. |
| 1988 | Allergan, Inc. acquired the rights to distribute Dr. Scott’s Botulinum Toxin Type A product, Oculinum. Allergan researches other medical uses of botulinum toxin. |
| 1989 | Allergan introduces the first botulinum toxin approved by the FDA to treat blepharospasm (eyelid spasms) and strabismus (crossed eyes). FDA approval to change the product’s name to BOTOX® (Botulinum Toxin Type A) |
| 2000 | FDA approves BOTOX® therapy for cervical dystonia to reduce the associated abnormal head position and neck pain. |
| 2002 | FDA approves BOTOX® Cosmetic (onabotulinumtoxinA), the same formulation as BOTOX®, with dosing specific to moderate to severe frown lines between the eyebrows (glabellar lines). |
| 2004 | FDA approves BOTOX® for hyperhidrosis (excessive underarm sweating) when antiperspirants don’t work well enough. |
| 2009 | 20-year anniversary of BOTOX®. |
| 2010 | FDA approves BOTOX® therapy for increased muscle stiffness in elbow, wrist, and finger muscles with upper limb spasticity. BOTOX® is the first medicine to be studied and then approved by the FDA specifically for the prevention of headaches in adults with Chronic Migraine who have 15 or more days each month with headache lasting 4 or more hours each day in people 18 years or older. |
Clinicbe® skin treatments, Belgravia:
Dr Barbara Kubicka is a highly experienced botox® practitioner working with clients in London from her Knightsbridge clinic.
Botox injections are one of the most popular treatments at Clinicbe®, who also offer a range of cosmetic and medical skincare treatments including fillers as well as mesotherapy and dermaroller. The clinic is also known for innovative treatments such as PRP (plasma rich therapy), PDT (photo dynamic therapy) and exclusive treatments include “Eyedealise”, “Neck and Jawline Programme” and the “Triple Synergy Hair System” for men and women who are suffering from hair loss.
Clinicbe® has a holistic approach to skincare, going beyond the immediate concerns of its patients to understand and treat the person as a whole. Dr Kubicka’s extensive range of cosmetic and medical skincare treatments is complemented by dietary advice and lifestyle/wellbeing services from fashion consultancy to counselling, fitness training and sports medicine.
For further information or to arrange a confidential consultation, please contact us:
Tel: 0207 125 0521
Address: Clinicbe®, Knightsbridge, London, SW1X
Email: info@clinicbe.com
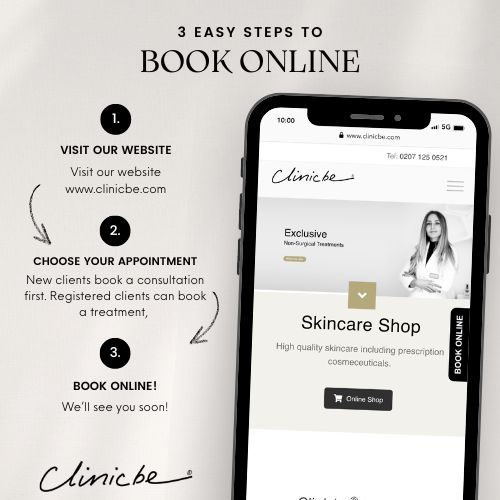
Website: www.clinicbe.com